Hey there,
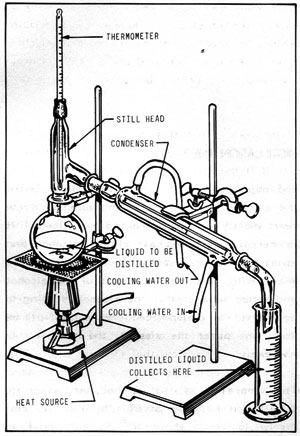
Chemists among you, I need a little bit of help. AS Level (UK) Evaluative coursework next week, and I need someone to explain to me the method used in preparing Pentan-2-One. I've looked online, searched plenty of different combinations, and have gone through my books and I've found nowt.
Any form of help would be greatly appreciated. =]
-Thanks
Chemists among you, I need a little bit of help. AS Level (UK) Evaluative coursework next week, and I need someone to explain to me the method used in preparing Pentan-2-One. I've looked online, searched plenty of different combinations, and have gone through my books and I've found nowt.
Any form of help would be greatly appreciated. =]
-Thanks